Hexamethylene diacrylate (1,6-Hexanediol diacrylate)
CAS:13048-33-4
MF:C12H18O4
MW:226.27
Chemical structure:
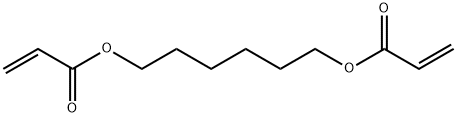
Synonyms:
HDODA,
Photomer 4017 T,
c716,
1,6-BIS(ACRYLOYLOXY)HEXANE,
1,6-HEXAMETHYLENE DIACRYLATE,
1,6-HEXANEDIOL DIACRYLATE,
HEXAMETHYLENE GLYCOL DIACRYLATE,
1,6-hexanedioldi-2-propenoate,
2-Propenoicacid,1,6-hexanediylester,
acrylicacid,hexamethyleneester,
Hexamethyleneacrylate,
Hexamethylenediacrylate,
Hexaneglycoldiarcylate,
Kayaradhdda,
Nkesterahd,
Hexanediol diacrylate (HDDA),
Properties:
Appearance:clear colorless to pale yellow
Physical State : Liquid
Melting Point : 5℃
Boiling Point : 107℃ at 0.2 mmHg
Flash point:113 ℃
Density : 1.01 g/cm3 at 25℃
Vapour pressure:<0.01hPa
Vapour density:7.81
Solubility:Slightly miscible with water.
Refractive Index:1.456-1.460
Application:
Hexamethylene diacrylate (also known as hexanediol diacrylate) is a di-functional acrylic monomer that can be polymerized by free radicals.
It is used in ultra violet (UV) and electron beam (EB) applications as a reactive component in formulating coatings and inks, furniture and floor coatings, coatings on plastic substrates, varnishes for packing items and more. urable lacquers and pigmented coatings, used in paper and board coatings, wood finishes vinyl flooring and flexible vinyl.
Further, it is used in adhesives, sealants, alkyd coatings, elastomers, photopolymers, and inks for improved adhesion, hardness, abrasion and heat resistance.
The hexamethylene diacrylate cross-linked polystyrene resin can be used for the solid phase synthesis of hydrophobic peptides
Package: 5kg/drum or 200kg/drum (blue plastic drum), and IBC drum with metal frame
Storage : Store at room temperature
Delivery
1) high temperature, direct sunshine and fire must be avoided when transported.
2) Not dangerous chemical and non-flammabl
Introduction and experiment of Hexamethylene diacrylate (1,6-Hexanediol diacrylate),cas:13048-33-4:
In the printing, coating and ink industries, photocurable systems are becoming increasingly popular and multi-functional acrylates are one of the most commonly used monomers due to their high reactivity (fast curing). In this paper, we use molecular dynamics and graph theory tools to investigate the thermo-mechanical properties and topology of hexanediol diacrylate (HDDA) polymer networks. The gel point was determined as the point where a giant component was formed. For the conditions of our simulations, we found the gel point to be around 0.18 bond conversion. A detailed analysis of the network topology showed, unexpectedly, that the flexibility of the HDDA molecules plays an important role in increasing the conversion of double bonds, while delaying the gel point. This is due to a back-biting type of reaction mechanism that promotes the formation of small cycles. The glass transition temperature for several degrees of curing was obtained from the change in the thermal expansion coefficient. For a bond conversion close to experimental values we obtained a glass transition temperature around 400 K. For the same bond conversion we estimate a Young's modulus of 3 GPa. Both of these values are in good agreement with experiments.
Reference:
(1)PubChem. "1,6-Hexanediol diacrylate". pubchem.ncbi.nlm.nih.gov. Retrieved 2020-02-12.
(2) "1,6-Hexanediol diacrylate | C12H18O4 | ChemSpider". www.chemspider.com. Retrieved 2020-02-12.
(3)Ajiboye, Gbenga (2012). "Industrially relevant epoxy-acrylate hybrid resin photopolymerizations".
(4)Abrasion Resistance Testing-Vehicle Exterior Graphics and Pin Striping, SAE International, doi:10.4271/j1847_198906
(5)"13048-33-4 - 1,6-Hexanediol diacrylate, 99% (reactive esters), stab. with 90ppm hydroquinone - HDODA - 43203 - Alfa Aesar". www.alfa.com. Retrieved 2020-02-27.
(6)Ohara, Takashi; Sato, Takahisa; Shimizu, Noboru; Prescher, Günter; Schwind, Helmut; Weiberg, Otto; Marten, Klaus; Greim, Helmut (2003). "Acrylic Acid and Derivatives". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a01_161.pub2.
(7) "Epoxy Polyacrylate Resins". www.hexion.com. Retrieved 2020-02-12.
(8)Malini, K.; Periyaraja, S.; Shanmugam, P. Morita-Baylis-Hillman reaction of pyridine-2, 3, and 4-carboxaldehydes and N-methyl isatin with mono and diacrylates: synthesis of highly functionalized pyridine ligands and isatin derivatives. Tetrahedron Lett. 2015, 56 (36), 5123-5127.
(9)Lee, H.; Lee, S. G.; Doyle, P. S. Photopatterned oil-reservoir micromodels with tailored wetting properties. Lab Chip 2015, 15 (14), 3047-3055.